ASEAN Today: Vietnam-produced COVID-19 vaccines help raise the country’s geopolitical status
 |
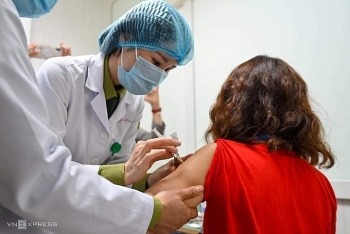
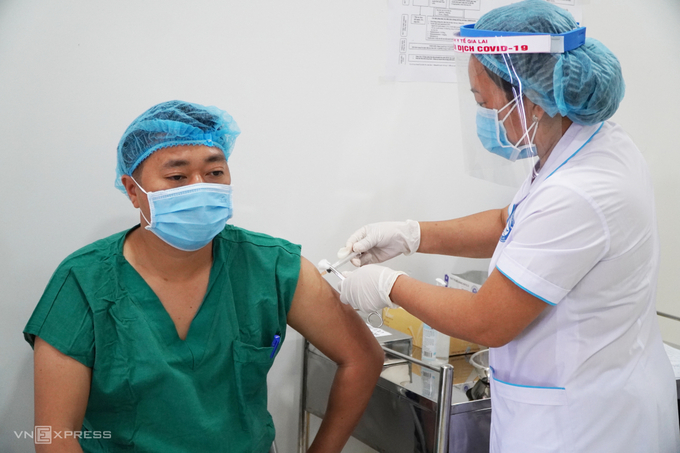
| Vietnam began human trials of homemade COVID-19 vaccine. Photo: Vnexpress |
Vietnam has pushed domestic vaccine development in part to reduce reliance on any single foreign source. But its willingness to share vaccine technology and support production abroad may help raise the country’s geopolitical status among the developing world, said ASEAN today.
Two Vietnamese vaccines have already become the first drugs developed in Southeast Asia to begin clinical trials.
Vietnam’s Nanogen Pharmaceutical Biotechnology has developed a candidate vaccine called Nanocovax, now in phase 2 clinical trials, which the company reports is effective at triggering an immune response and will likely protect against the new strain of coronavirus initially detected in the UK. If the current trials are a success, the government could move to issue emergency approval and Nanocovax could be in use as early as May, according to Nikkei. A second vaccine known as Covivac, developed by the Institute of Vaccines and Medical Biologicals (IVAC), began clinical trials on March 15.
As the majority of vaccine doses go to the United States and Europe, most drug companies are unwilling to share their technology and many countries can’t afford to purchase the necessary doses. Like many governments, Vietnam is also wary of becoming over reliant on any single country for its vaccines.
But Vietnam’s vaccine push is about more than balancing foreign influence. Vietnam’s success in containing COVID-19—the country has seen only 35 deaths—has prompted speculation around the world about its rising status. Vietnam’s vaccines may allow the government to position itself as a leader among developing countries by supporting their access to the drugs and their recovery as a whole. Hanoi’s approach to vaccine policy points to Vietnam’s changing geopolitical role.
By supporting vaccine development and production in the Global South, Vietnam aligns itself with the push for global vaccine equity. Where drug companies in the US and the UK are firmly attached to their patents and restrict production overseas, Hanoi would offer countries a cheap, reliable and politically neutral vaccine alternative.
 |
Vietnam stakes its place in Southeast Asia’s vaccine struggles
Vietnam has already begun inoculations with the first of 30 million doses of the AstraZeneca vaccine and has received a small amount of the Sputnik V vaccine donated from Russia. The government has also sought vaccines from Pfizer in the US, saying it aims to secure a total of 150 million doses by the end of the year, enough to vaccinate 70% of the population.
Thailand is also developing two of its own vaccines, one by Chulalongkorn University and one by Mahidol University. The first doses of the Chulalongkorn vaccine are being produced by partners in California, though domestic manufacturers aim to be ready to produce around 5 million doses per year by the end of 2021. By comparison, Vietnam’s Nanogen says it already has capacity to manufacture 10 million doses per year.
Other Southeast Asian governments are hoping to at least begin domestic production of foreign-developed vaccines. Indonesia, Thailand and Malaysia are all working with foreign vaccine producers, chiefly Sinovac or AstraZeneca, to manufacture or finish the drugs in-country. A medical school in Singapore is also developing a single-dose vaccine in partnership with US firm Arcturus Therapeutics.
Vietnam has signaled that it hopes to work with countries in the region and beyond to help them produce Vietnamese vaccines in-house. Nanogen says it will share its vaccine production process and technology with manufacturers in other countries.
“We are willing to transfer our techniques. The problem is [whether] they can produce it or not,” said Do Minh Si, Nanogen’s director of research and development.
The company was first tapped by the government to develop monoclonal antibody therapy for COVID-19 treatment, but there weren’t enough cases of the disease in the country to conduct clinical trials. It soon pivoted to vaccine development, joining the race towards domestic production. Along with Nanocovax and Covivac, there are also two Vietnamese vaccines that have yet to begin trials: Vabiotech, developed by the Vaccine and Biological Production Company No. 1, and Polyvac, developed by the Center for Research and Production of Vaccines and Biologicals.
Vietnam’s efforts are encouraging to those advocating for more equitable access to vaccines, as vaccinations in the US and other wealthy countries are a year or more ahead of the rest of the world.
“Whilst rich nations are vaccinating one person a second, the majority of poor countries have yet to give a single shot,” said Oxfam Head of Inequality Policy Max Lawson. “Not only are vaccines far too expensive, [but] poor countries are being forced to the back of the queue by rich nations, and patents and monopolies of pharmaceutical corporations are artificially rationing supply.”
 | Covid-19 vaccine supplied by Covax to arrive in Vietnam later than expected More than 4 million doses of Covid-19 vaccine supplied by global vaccine access mechanism Covax would arrive in Vietnam later than the scheduled time of ... |
 | Vietnam Airlines to pilot digital health passport project National flag carrier Vietnam Airlines will work with domestic and foreign agencies to roll out a “COVID-19 vaccine passport” project. |
 | Thailand begins vaccine trials on human in March Thailand started human trials on Monday (Mar 22) of a domestically developed COVID-19 vaccine and expects to deploy it next year. |
Recommended
 Viet's Home
Viet's Home
French-Vietnamese Author Introduces Book Series "Memories of Overseas Vietnamese"
 Viet's Home
Viet's Home
Vietnam's Human Development Index Remains High
 Viet's Home
Viet's Home
Vietnam’s Mark on UN Day of Vesak Celebrations
 Viet's Home
Viet's Home
Art Program Spreads Message of Peace Worldwide
 Expats in Vietnam
Expats in Vietnam
Look Forward to New Developments in Vietnam - US Relations
 Viet's Home
Viet's Home
She Feeds the World: 8,000 Individuals Adopt More Sustainable Agricultural Practices
 Viet's Home
Viet's Home
Over 200 Valuable Documents Displayed at 'Mountains and Rivers Connected One Strip' Exhibition
 Viet's Home
Viet's Home