Thailand begins vaccine trials on human in March
| Indonesia halts the use of AstraZeneca's COVID-19 after Thailand, in fear of blood clots | |
 |
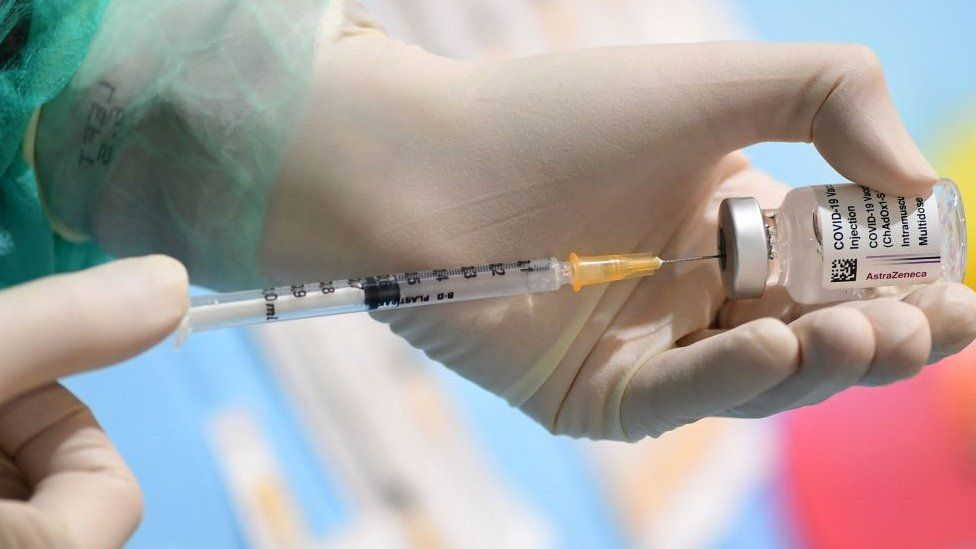
| Public Health Minister Anutin Charnvirakul arrives at a meeting to discuss the management of the coronavirus disease (Covid-19) outbreak and vaccines in Bangkok, Jan 25, 2021. Photo: Reuters |
Thailand will begin clinical trials of a locally developed Covid-19 vaccine in March, making it the nation’s first inoculation program to enter human trials.
Thai vaccine human trials to begin in March
The program, led by the Government Pharmaceutical Organisation, Mahidol University and the Programme for Appropriate Technology in Health, started its research in mid-2020 and its animal testings showed good results, Health Minister Anutin Charnvirakul said on Wednesday, according to Bang Kok Post.
“Thailand isn’t just relying on one source for vaccines. We’re actively looking for ways to procure the shots, including producing them ourselves,” Mr Anutin said, adding that if the human trials are successful, the state drug manufacturer will be able to produce 25 to 30 million doses of the vaccines per year.
“Even though we can produce vaccines in the country, it is from technology transfer and under the management of the owners of the brands and technology. But today, if we are successful we can be self-reliant and determine our own direction.”
A total of 460 volunteers would be recruited for the human trials. They must be in good health and free of Covid-19. Volunteers will be screened and have their backgrounds checked and undergo health and blood checks.
Punnee Pitisuttithum, the head of the Vaccine Trial Centre at Mahidol University’s Tropical Medicine Department, said that the first human trial was conducted on 4 volunteers yesterday. Banchong Mahaisavariya, president of Mahidol University, said the first and second phases of human trials will test the safety of the vaccine and its ability to trigger immunity, said The Thaiger.
The GPO is aware of SARS-CoV-2 virus mutations so the vaccine is also being developed to help with this situation, adding that the pilot vaccine showed it was effective against the South African strain.
Chulalongkorn University is also developing its own vaccine, which uses Messenger RNA technology with human trials expected to begin soon.
Thailand has recorded 27,876 coronavirus cases in total, with 91 deaths.
| An additional homegrown vaccine is under development by Chulalongkorn University which uses Messenger RNA (mRNA) technology and is expected to start human trials soon, The Phuket News reported. Amid tight global supplies and concerns around certain vaccines already available, certain countries such as Japan and Taiwan have turned to develop their own; Vietnam expects its home-developed vaccine to be available before the end of this year. |
Iran is the first country to begin the human trial of locally made virus vaccine
 |
| Tayebeh Mokhber is injected with the Coviran coronavirus vaccine produced by Shifa Pharmed, part of a state-owned pharmaceutical conglomerate, in a ceremony in Tehran, Iran, Tuesday, Dec. 29, 2020. The first study of the safety and effectiveness of the coronavirus vaccine in Iran began Tuesday, state TV reported, with dozens due to receive the domestically developed shot in the hardest-hit country in the Middle East. Photo: AP News |
The first study of the safety and effectiveness of a coronavirus vaccine in Iran began Tuesday, state TV reported, with dozens due to receive the domestically developed shot even as details about its production remained slim.
The vaccine, the first in the country to reach human trials, is produced by Shifa Pharmed, part of a state-owned pharmaceutical conglomerate known as Barekat. The company’s website describes it as involved in the large-scale production of antibiotics and penicillin, without offering any details about its coronavirus research, results of animal trials or previous vaccine development since its founding in 1995.
The study, a Phase 1 clinical trial, will enroll a total of 56 volunteers to receive two shots of the Iranian vaccine within two weeks, according to Hamed Hosseini, a clinical trial manager. Results are to be announced roughly a month after the second shot. Three people received the first injections on Tuesday in a ceremony at a Tehran hotel attended by the country’s health minister. State TV announced that none of the injections had so far caused any “fevers” or “bodily shocks.”
President Hassan Rouhani has said Iran is cooperating with a “foreign country” to produce another vaccine expected to run in tests in human volunteers in February, without offering further details.
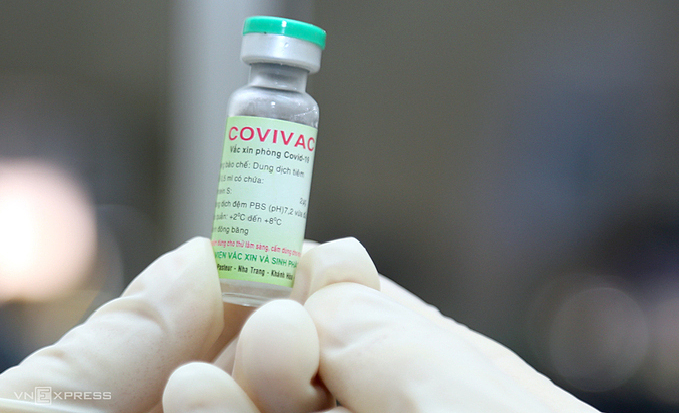
| In Pictures: Made-in-Vietnam Covivac vaccine's human trial procedures All volunteers get general health checks, have their blood samples and pregnancy test taken before getting the indigenous Covivac vaccine Monday morning at Hanoi Medical ... |
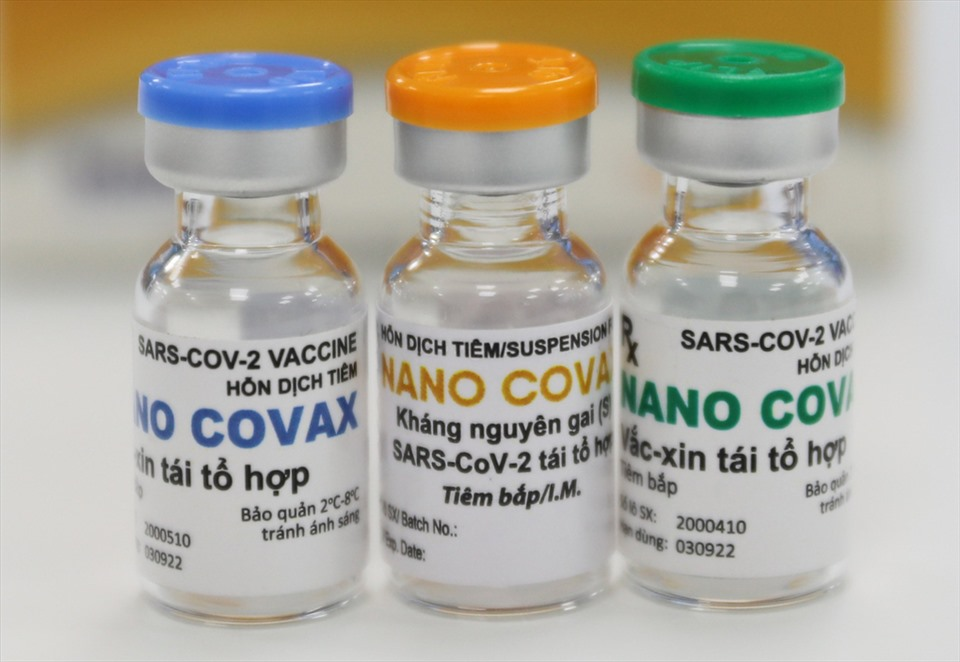
| Many elderly people participate in phase two human trials of Made-in-Vietnam Covid-19 vaccine A total of 367 volunteers, including 30 people over the age of 60, have received Nano Covax, the first Made-in-Vietnam Covid-19 vaccine as part of ... |
| Vietnam recruits volunteers for phase one human trials of 2nd homegrown Covid-19 vaccine The Institute of Vaccines and Medical Biologicals (IVAC) is recruiting volunteers for the first phase of human trials of Covivac, the second Made-in-Vietnam Covid-19 vaccine. |
Recommended
 World
World
India sending Holy Relics of Lord Buddha to Vietnam a special gesture, has generated tremendous spiritual faith: Kiren Rijiju
 World
World
Why the India-US Sonobuoy Co-Production Agreement Matters
 World
World
Vietnam’s 50-year Reunification Celebration Garners Argentine Press’s Attention
 World
World
"Will continue offering our full support to Indian govt": US FBI Director after Pahalgam attack
Popular article
 World
World
"Great Leader": JD Vance Lauds PM Modi During His India Visit
 World
World
Trump’s Tariff Pause: A Strategic Move from “The Art of the Deal”?
 World
World
"Indian Navy's participation in AIKEYME exercise matter of great happiness": Admiral Dinesh Kumar Tripathi
 World
World