Remdesivir approved for COVID-19 treatment in Japan
| Massive Coronavirus drugs production face challenges due to high cost of manufacturing | |
| Top coronavirus drugs and vaccines in development | |
| Latest treatments and drugs for Coronavieus (COVID-19) |
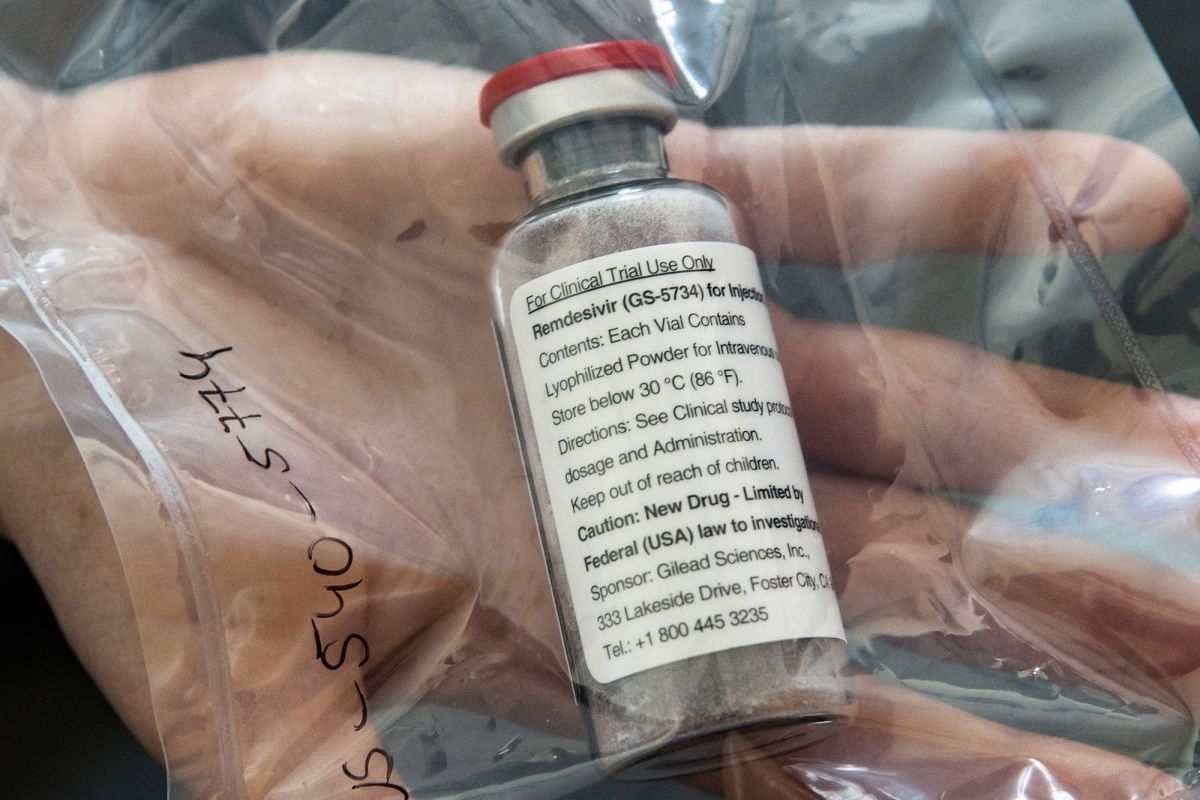 |
| Remdesivir (Photo: AFP) |
Japan, however, as yet does not know when it will get its first doses of remdesivir or how much, the health ministry official said.
The country reached the decision just three days after the U.S. drugmaker filed for fast-track approval for the treatment.
“There has so far been no coronavirus medicine available here so it is a significant step for us to approve this drug,” Reuters quoted a Japanese health ministry official said at a press briefing. Remdesivir will be give to patients with severe COVID-19 symptoms, he added
Prime Minister Shinzo Abe said last week the government was getting ready to give a speedy green light to the experimental drug developed by U.S. firm Gilead Sciences, Japan Today reported.
The U.S. go-ahead came after a major clinical trial showed remdesivir -- originally developed to treat Ebola -- shortened the time to recovery in some patients by a third.
Japan, with just over 16,000 infections and under 800 deaths, as of May 8, has recorded fewer cases than other major industrialized nations.
However, a steady rise in cases has put pressure on medical facilities in some parts of the country, and a drug that helps patients recover more quickly could help in freeing up hospital beds.
On Monday, Japanese Prime Minister Shinzo Abe extended a month-long state of emergency until the end of May in an attempt to slow the spread of the coronavirus.
With no other approved treatments for COVID-19, interest in remdesivir, the first drug shown to help fight COVID-19, is growing around the world. Administered by intravenous infusion, it was granted authorisation last week by the U.S. Food and Drug Administration for emergency use for the disease caused by the novel coronavirus.
Gilead says the drug has improved outcomes for people suffering from the respiratory disease and has provided data suggesting it works better when given in the early stages of infection.
| Earlier on May 1, US FDA allows emergency use of remdesivir drug for coronavirus. Following the allowance, the drug is now be administered to patients with severe conditions, such as those experiencing breathing problems requiring supplemental oxygen or ventilators, according to ITV News. |
 |
| (Photo: CNBC) |
As for Avigan, developed by Japanese firm Fujifilm Toyama Chemical, Suga said the government "aims to approve it this month" if a clinical trial involving 100 patients proves effective.
The drug, whose generic name is favipiravir, was approved for use in Japan in 2014 but only in flu outbreaks that are not being effectively addressed by existing medications.
It is not available on the market and can only be manufactured and distributed at the request of the Japanese government.
Favipiravir, which can be taken orally as a pill, works by blocking the ability of a virus to replicate inside a cell.
Remdesivir incorporates itself into the virus's genome, short-circuiting its replication process.
Avigan has been shown in animal studies to affect fetal development, meaning it is not given to pregnant women, Japan Today reported.
 | Japanese Ambassador praises Vietnam for its COVID-19 response Ambassador Yamada expressed his admiration for Vietnam’s effective COVID-19 fight and said that was an exemplary response to the pandemic. |
 | Repatriated student creates amazing COVID-19 isolated -day sketches to raise charity funds "I'm home" - a book gathering paintings drawn by Tang Quang during the period in the central quarantine at the Zone 7 military school in ... |
 | COVID-19 update: Coronavirus found in sperm samples, Chinese doctors say The new coronavirus can persist in men's semen even after they have begun to recover, a finding that raises the possibility the virus could be ... |
Recommended
 World
World
Pakistan NCRC report explores emerging child rights issues
 World
World
"India has right to defend herself against terror," says German Foreign Minister, endorses Op Sindoor
 World
World
‘We stand with India’: Japan, UAE back New Delhi over its global outreach against terror
 World
World
'Action Was Entirely Justifiable': Former US NSA John Bolton Backs India's Right After Pahalgam Attack
 World
World
US, China Conclude Trade Talks with Positive Outcome
 World
World
Nifty, Sensex jumped more than 2% in opening as India-Pakistan tensions ease
 World
World
Easing of US-China Tariffs: Markets React Positively, Experts Remain Cautious
 World
World