Politburo gives nod to COVID-19 vaccine import for domestic use
 |
| Party General Secretary and State President Nguyen Phu Trong (Second from left) presides over the first meeting of the Political Bureau and Secretariat of the Communist Party of Vietnam. Photo: VNA |
The 13th Political Bureau, the supreme policy-making body of the Communist Party of Vietnam (CPV), along with the CPV Secretariat, approved the Government’s plan to purchase COVID-19 vaccines for domestic use, at their first meeting held on February 18 in Hanoi.
The decision has been taken that funds for buying, transporting, preserving, and using the vaccines will mainly come from the State budget and other sources, VOV reported.
The purchase and use of vaccines carried out in emergency cases must also comply with procurement regulations regarding special cases and meet the recommendations of the World Health Organization (WHO) and vaccine manufacturers.
The Politburo and the Secretariat also assigned the Government’s Party Affairs Board to direct the Government, relevant ministries, and agencies to competently organise the purchase and use of COVID-19 vaccines. This will be done whilst simultaneously promoting the local research and production of COVID-19 vaccines in an effort to meet international standards and adapt to new emerging variants of the SARS-CoV-2 virus.
 |
| The total cases of COVID-19 in Vietnam reached 2,347 as of February 18 evening, including 755 locally-transmitted cases since January 27. Photo: VNA |
With regard to the key tasks ahead in the first quarter of the year, the Politburo and the Secretariat requested that Party organisations at all levels concentrate on COVID-19 control measures to be done in a way that does not impact people’s lives and economic development.
The Politburo and the Secretariat also decided to raise the COVID-19 alerts to the highest level whilst mobilising the entire political system to get involved in epidemic prevention measures and control from the beginning of the year.
Since the initial outbreak of the SARS-CoV-2 virus three weeks ago, cases have since spread to 13 cities and provinces, with as many as 737 new infections being detected nationwide, the majority of which are in Hai Duong province, the country’s largest COVID-19 hotspot.
While locally-produced vaccines are in the phase of human clinical trials, the Government has also agreed to import vaccines for domestic use. More than 200,000 doses of the AstraZeneca vaccine are anticipated to arrive in Vietnam on February 28.
This will be the first batch of a total of 30 million doses which the country will receive after reaching a deal to purchase from AstraZeneca.
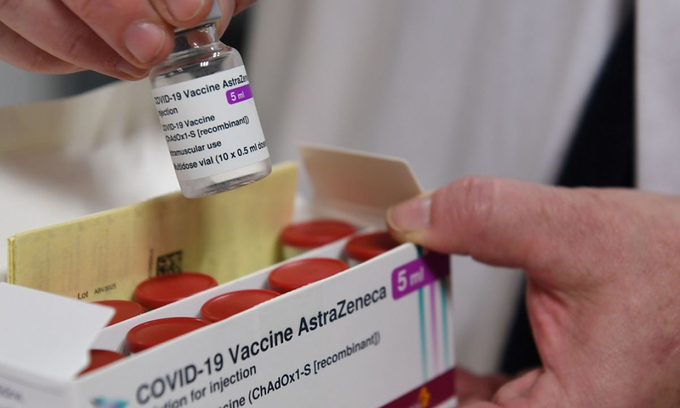 |
| A health worker opens a box containing the Oxford/AstraZeneca Covid-19 vaccine in France. Photo: AFP. |
Currently, this vaccine is licensed for one-year circulation or conditional import in the Philippines, Thailand, the U.K., Vietnam and some other countries to help fight COVID-19, according to VnExpress.
The ministry earlier said doctors and other staff in frequent contact with Covid-19 patients or suspected infectees will be among the first to be vaccinated.
Other people on the priority list will be the elderly and those with chronic diseases that make them more vulnerable if they get infected, and officials in the diplomatic service.
Prime Minister Nguyen Xuan Phuc said importing Covid-19 vaccines should be a foremost priority of the government, while calling for quick research progress on domestic Covid-19 vaccines as well.
Vietnam is also expected to receive 4.9 million vaccine doses by the end of this month, provided via Covax, a global mechanism for developing, manufacturing and procuring COVID-19 vaccine candidates and help member countries access vaccines as they become available.
Vietnam has four domestic Covid-19 vaccines developed by Nanogen Pharmaceutical Biotechnology JSC, the Institute of Vaccines and Medical Biologicals, the Vaccine and Biological Production Company No. 1 and the Center for Research and Production of Vaccines and Biologicals.
Nanocovax, produced by Nanogen Pharmaceutical Biotechnology JSC, has finished its first phase of human trials and is expected to begin the second one this month. Covivac of the Institute of Vaccines and Medical Biologicals is expected to commence human trials this month as well./.
| The total cases of COVID-19 in Vietnam reached 2,347 as of February 18 evening, including 755 locally-transmitted cases since January 27. On February 18, as many as 25 patients were given the all clear, raising the total number of recoveries to 1,605. The death toll remains at 35. Some 144,070 people who had close contact with COVID-19 patients or arrived from pandemic-hit areas are being quarantined nationwide. Hai Duong - Vietnam's current largest COVID-19 hotspot - had basically managed to put under control five big outbreaks by February 18, said the provincial steering committee for COVID-19 prevention and control. |
 | First batch of 204,000 Covid-19 vaccine doses to arrive in Vietnam on Feb 23 The Ministry of Health has approved the import of 204,000 doses of the Oxford/AstraZeneca Covid-19 vaccine to battle the fresh wave of the coronavirus in ... |
 | Pandemic-hit Hai Duong province asks for assistance as Covid-19 cases surge Given the complicated development of the Covid-19 outbreaks, the northern province of Hai Duong has requested the Ministry of Health to assist it in establishing ... |
 | Pandemic-hit northern province offers Covid-19 testing on demand Starting February 16, six medical facilities in the Northern Quang Ninh province, the country’s current Covid-19 hotspot offers Covid-19 testing on demand at a maximum ... |
Recommended
 National
National
Vietnam News Today (Jun. 4): Vietnam - Promising Candidate for Southeast Asia’s Next Powerhouse
 National
National
Shangri-La Dialogue 22: Vietnam Highlights Some Issues of Ensuring Stability in a Competitive World
 National
National
Vietnam News Today (Jun. 3): PM Pham Minh Chinh to Attend UN Ocean Conference, Visit Estonia, Sweden
 National
National
Vietnam News Today (Jun. 2): Vietnamese Trade Mission Sounds Out Business Opportunities in United States
 National
National
Vietnam News Today (Jun. 1): Vietnamese, Japanese Firms Foster Partnership
 National
National
Vietnam News Today (May 31): Vietnam Strongly Supports Laos’s National Development
 National
National
Vietnam News Today (May 30): Vietnam, Venezuela Reinforce Ties Through People-to-people Diplomacy
 National
National





