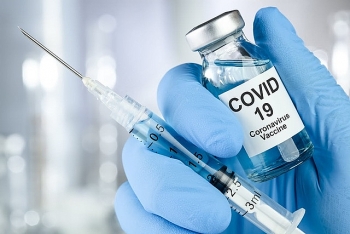
China approves inhaled CanSino vaccine for clinical trials
 |
| CanSino said their clinical trial application got the green light, but also noted that the vaccine's safety and efficacy remain " subject="" to="" confirmation"="" in="" trials.="" photo:="" reuters |
China's leading vaccine producer CanSino Biologics obtained the national drug regulator's approval on Monday to start clinical trials of its latest recombinant COVID-19 vaccine (Adenovirus Type 5 Vector) in inhalation form.
Inhaled CanSino vaccines to be improved
The move comes after the National Medical Products Administration gave another CanSino vaccine conditional approval last month, allowing it for public use.
China currently has five coronavirus vaccines that have been given conditional market approval or allowed for emergency use, but none of these are administered by inhalation.
CanSino said in its latest filing on the Hong Kong stock exchange that the vaccine for inhalation was jointly developed by the company and the Beijing Institute of Biotechnology, adding that their clinical trial application got the green light on Monday, according to The Strait Times.
The Global Times learned that the vaccine, which was developed in cooperation with the Institute of Military Medicine under the Academy of Military Sciences, is no different in terms of virus species, cell banks, production technology or preparation formula from the company's single-dose Ad5-nCoV COVID-19 vaccine, which is already in use - it just has an additional atomization inhalation device.
The inhaled vaccine can stimulate an immune response in the mucous membranes of a person's respiratory tract. This version is less painful and more accessible, especially for children and vulnerable people, experts said.
The vaccine can be atomized into tiny particles in a device and then inhaled into the respiratory tract and lungs to stimulate mucosal immunity, a Beijing-based immunological expert who prefers to be not identified told the Global Times.
Research has confirmed that respiratory droplets and close contact are still the main transmission routes of the novel coronavirus.
Mucosal immunity acts as the first barrier against the virus and is essential for the prevention of both mild and severe cases, Jiang Chunlai, a professor at Jilin University's School of Life Sciences, told the Global Times.
Beijing makes Chinese jabs compulsory in a bid to lead the vaccine race
 |
| Photo: Global Times |
As of March 20, China had administered 74.96 million coronavirus vaccine doses, according to Our World in Data, a collaboration between Oxford University and a charity.
Chinese embassies in some countries, including the United States, Australia and India, have issued notices saying the country will open visa applications to select people who have taken a China-made jab, said Medical Express.
On Tuesday, a Chinese Foreign Ministry official said it is in "close communication" with various countries and "willing to reach mutually beneficial arrangements" to facilitate cross-border travel.
After shutting its borders more than a year, China is opening up again but only for those who take Chinese vaccines.
Beijing has come up with two sets of rules: those who take Chinese vaccines will be given "conveniences" and those who don't will have to fill up more forms, go through tests, and once you land in China, spend up to three weeks in a quarantine even if you have been vaccinated by a non-Chinese shot.
The interesting point to note here is that all these foreign vaccines have proven their efficacy while the Chinese vaccines are unproven.
Chinese embassies have issued similar notices in 20 countries such as Australia, South Korea, and the US. The interesting thing to note here is that none of these countries have approved any Chinese vaccines against coronavirus.
However, that hasn't stopped China from injecting people with these shots. Since last year, China has administered tens of thousands of people with its vaccines and now it wants to expand its market, so it is coming up with these discriminatory rules.
| If you do survive the Chinese shot, you also get a simplified application process as you will need fewer documents to apply for a visa. You would also not be asked to procure an invitation letter. Earlier, if you traveled to China for business purposes, you had to produce an invitation letter which was given by Chinese government departments. Now, a statement issued in Germany suggested that the requirement will be waived off if you take a Chinese vaccine, WION reported. |
| ASEAN Today: Vietnam-produced COVID-19 vaccines help raise the country’s geopolitical status ASEAN Today, a leading ASEAN commentary site on March 24 ran an article titled “Vietnam’s in-house vaccines will play a role in pandemic geopolitics”, highlighting ... |
 | First batch of vaccine under Covax to arrives in Vietnam next month More than 811,000 doses of the AstraZeneca COVID-19 vaccine will be delivered to Hanoi next month via the WHO-led Vaccines Global Access (Covax) initiative. |
 | Nearly 38.000 Vietnamese receive Covid-19 vaccine, more 3 provinces launch inoculation campaign this week As of March 23, more 1.829 people in Vietnam were administered the Covid-19 vaccine, raising the tally of recipients to 37.911. Three provinces of Quang ... |
Recommended
 World
World
Pakistan NCRC report explores emerging child rights issues
 World
World
"India has right to defend herself against terror," says German Foreign Minister, endorses Op Sindoor
 World
World
‘We stand with India’: Japan, UAE back New Delhi over its global outreach against terror
 World
World
'Action Was Entirely Justifiable': Former US NSA John Bolton Backs India's Right After Pahalgam Attack
Popular article
 World
World
Nifty, Sensex jumped more than 2% in opening as India-Pakistan tensions ease
 World
World
Easing of US-China Tariffs: Markets React Positively, Experts Remain Cautious
 World
World
India strikes back at terrorists with Operation Sindoor
 World
World