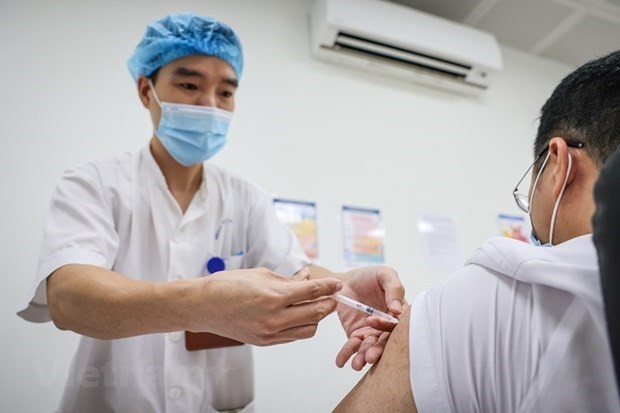
Vietnam adopts many measures to ensure Covid-19 AstraZeneca vaccination safety
| Three prioritized groups receiving Covid-19 vaccine in second phase in HCMC | |
| Health Ministry changes Covid-19 vaccine allocation plan | |
| Ho Chi Minh City receives more than 56,000 Covid-19 vaccine doses |
Responding to the press on concerns related to AstraZeneca vaccination on April 19 afternoon, Deputy Director of the National Institute of Hygiene and Epidemiology Duong Thi Hong affirmed “Vietnam always place the safety in parallel with increasing coverage of vaccines for its citizens”.
According to Hong, Vietnam’s health sector has adopted many measures to ensure the safety for Covid-19 AstraZeneca recipients. Firstly, all recipients have their health screened before administration. “We use the post-vaccination response monitoring system, part of the National Expanded Program on Immunization, which has been implemented methodically for more than 10 years”, Hong was quoted by Vnexpress as saying.
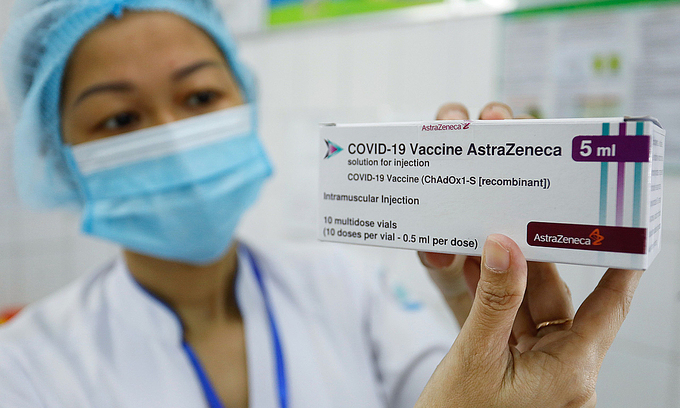 |
| A medical worker holds up a Covid-19 vaccine dose by AstraZeneca at the National Hospital for Tropical Diseases in Hanoi, March 8, 2021. Photo: VnExpress |
The system records post-vaccination responses daily. “It receives reports from all localities across the country on the number of people injected as well as the number of people displaying responses after inoculation. On this basis, we can grasp the safety level of the injection”, Hong added.
All medical workers are trained on how to use vaccines and how to treat cases with reactions by experts in clinical and emergency resuscitation. "Vaccination in Vietnam is always companied by a treatment system”, Hong said.
Although Vietnam to date hasn’t yet reported any case of blood clotting after inoculation, the Ministry of Health has accelerated to develop guidelines on vaccination safety, Hong said.
In addition, Vietnam has implemented an e-health record system so that vaccine recipients can report their situation after inoculation. The health facilities will synthesize and analyze data on this system daily.
As of April 19, more than 80.000 Vietnamese have received the AstraZeneca Covid-19 vaccine.
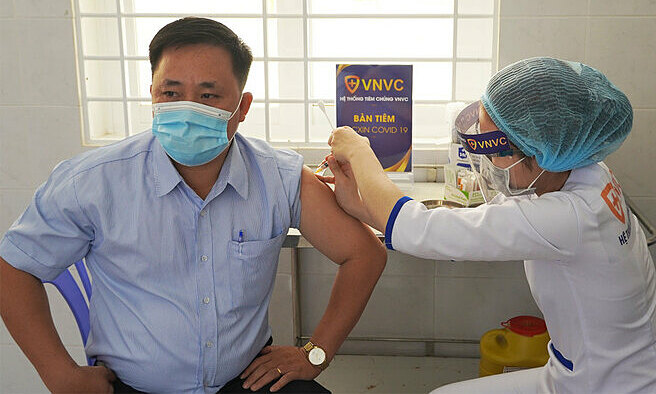 |
| A medical staff receives the AstraZeneca Covid-19 vaccine. Photo: VnExpress |
There have been 33% of recipients displaying commonly mild reactions such as swelling at the injection site, fatigue, mild fever, headache, and nausea. These reactions are normal which are also detected in other types of vaccines against measles, whooping cough, or tetanus.
There has been 1% of recipients experiencing severe reactions; however, they were timely treated and now are all in stable health conditions.
The post-injection reaction rate in Vietnam is lower than that announced by the producer.
Denmark previously abandoned the AstraZeneca Covid-19 vaccine after two recipients in the country suffered severe blood clots, one of which was fatal.
The World Health Organization (WHO) and the European Medicines Agency (EMA) said blood clots were very rare side effects of the AstraZeneca vaccine, adding that the vaccine's benefits far outweigh its risks. Thus, countries should continue to use it.
Vietnam received 117,600 AstraZeneca vaccine doses through its order from the producer in late February and another 811,200 doses through Covax on April 1. The ministry has distributed vaccine doses to 28 cities and provinces.
The country aims to secure around 150 million vaccine doses to cover 70 percent of its population by next year.
 | Ha Tinh prioritized individuals get Covid-19 vaccine on live platform Central province Ha Tinh on April 15 got a live broadcast while administering the AstraZeneca vaccine 46 medical staff. The live session is streamed to ... |
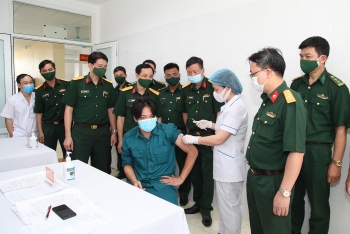 | Over 420 officers, soldiers in Quang Ninh get prioritized Covid-19 vaccine 424 officers and soldiers serving the coming 6th Vietnam-China Border Defense Friendship Exchange event were injected with AstraZeneca dose on April 15. |
 | Vietnam's homemade vaccine Nanocovax to enter third human trial phase in May The third clinical trial stage of made-in-Vietnam Covid-19 vaccine Nanocovax is scheduled to start on May 5, three months earlier than expected. |
Recommended
 National
National
Vietnam News Today (May 14): US Businesses Affirm Long-Term Commitment to Vietnam
 National
National
Vietnam News Today (May 13): Vietnam Maintains High Human Development Index Despite Global Slowdown
 National
National
Vietnam News Today (May 12): Party General Secretary Meets With Russian Experts, Intellectuals
 National
National
Vietnam News Today (May 11): Vietnam, Austria to Boost Cooperation in High-Tech Development, Innovation
Popular article
 National
National
Vietnam News Today (May 10): Vietnamese Peacekeepers Honored with UN Medal in South Sudan
 National
National
Vietnam News Today (May 9): Vietnam Ready to Work With Russia to Elevate Relations
 National
National
Vietnam News Today (May 8): Vietnam Remains Committed to UNCLOS
 National
National