Vietnam to launch first homegrown COVID-19 vaccine in September
 |
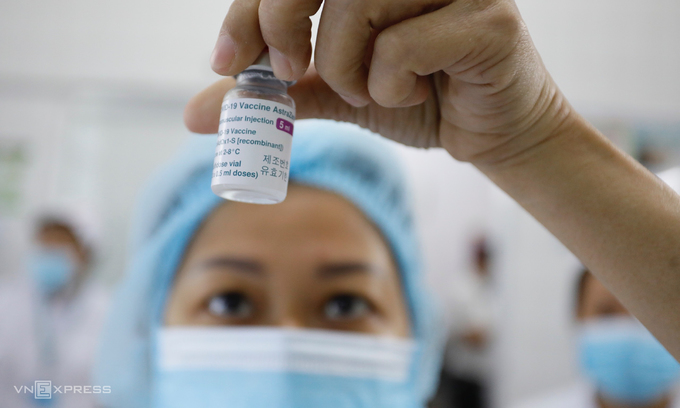
| NanoCovax is the first COVID-19 vaccine of Vietnam to be tested in clinical trials. Photo: VnExpress |
Vietnam expects to introduce its first domestically-developed COVID-19 vaccine in September, heard a meeting of the National Steering Committee for COVID-19 Prevention and Control on March 22.
It was reported at the meeting that Vietnam has four domestic COVID-19 vaccines developed by Nanogen Pharmaceutical Biotechnology JSC, the Institute of Vaccines and Medical Biologicals, the Vaccine and Biological Production Company No. 1 and the Center for Research and Production of Vaccines and Biologicals.
NanoCovax, developed by the Nanogen Pharmaceutical Biotechnology JSC, is the first COVID-19 vaccine of Vietnam to be tested in clinical trials.
The company has completed the first phase of human trials of its Nano Covax vaccine and began the second phase on February 26.
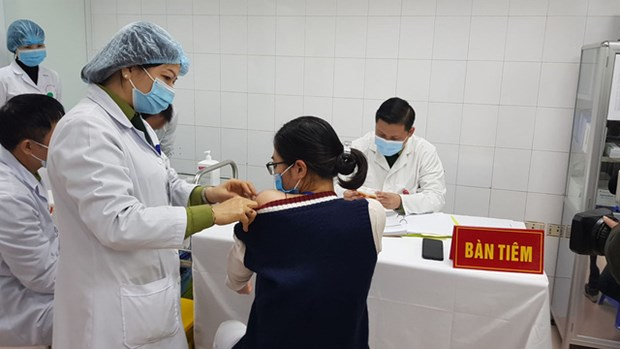 |
| The highest dose, 75mcg, of Nanocovax was given to three volunteers in Hanoi on January 12. Photo: MoH |
The first-stage trials of NanoCovax showed that the vaccinated volunteers have had antibodies against the UK variant (B117). Phase 2 focuses on seeking antibodies against the UK and South Africa variants.
A preliminary report on testing results is scheduled to be issued at the end of April.
The third phase of human trials is expected to begin in early May, involving 10,000-15,000 people in both Vietnam and other countries in Asia with severe coronavirus outbreaks.
Experts hope that the third phase would be completed at the end of the third quarter of this year, three months ahead of schedule.
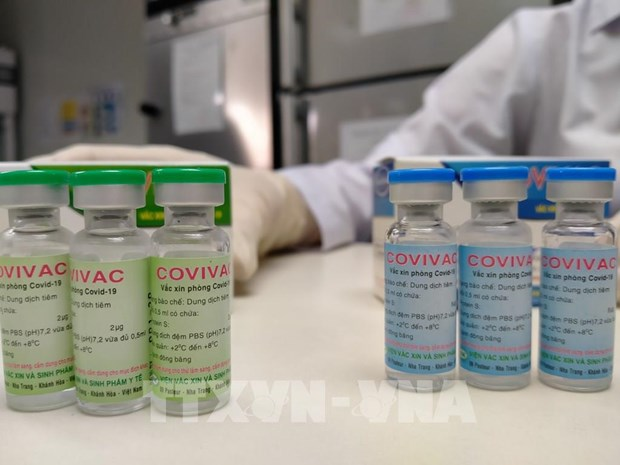 |
| COVIVAC is the second made-in-Vietnam candidate vaccine tested on humans. Photo: VNA |
Developed by the Institute of Vaccines and Medical Biologicals (IVAC) since last May, COVIVAC is the result of cooperation between IVAC and a number of partners, including the Hanoi Medical University, the Icahn School of Medicine at Mount Sinai in New York City, the University of Texas in Austin, and the US-based health organisation PATH.
Using primary chicken embryo cell culture - a technique the institute used previously to successfully produce seasonal flu vaccines - it is the second made-in-Vietnam candidate vaccine tested on humans.
Human trials on the second Vietnam-produced COVID-19 vaccine started on March 15.
A report on the outcomes of the first phase is expected to be completed in July. If the vaccine meets standards on safety and effectiveness, the second phase will be carried out at the medical centre in Thai Binh province’s Vu Thu district with a larger number of volunteers.
Meanwhile, the first phase of human trials of the vaccine developed by Vaccine and Biological Production No. 1 (VABIOTECH) is set to begin in April.
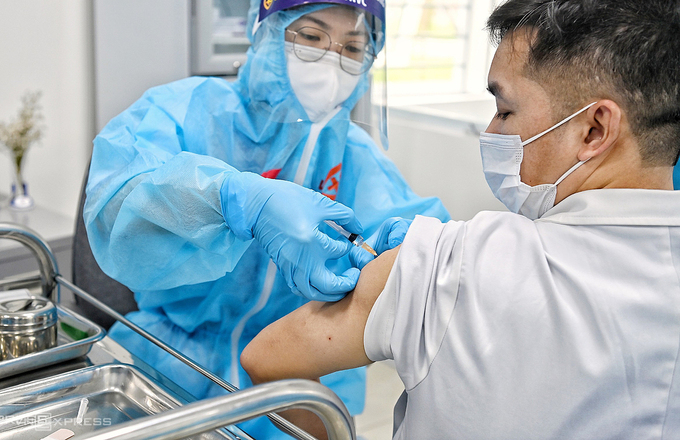
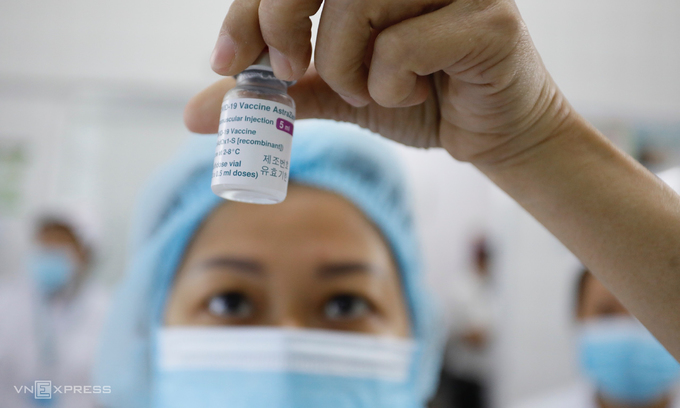
Regarding the AstraZeneca vaccine rollout, Vietnam has administered the vaccine to nearly 34,000 people who are frontline health workers and members of community-based anti-COVID-19 groups and the steering committees for COVID-19 prevention and control./.
 | How Vietnam implements 'Covid-19 vaccine passport'? Under the plan, after receiving the Covid-19 vaccine, recipients will be granted the vaccination certificates and QR-Codes. |
 | Overseas Vietnamese feel ‘more secure' after getting COVID-19 vaccine Some Vietnamese nationals in the US and Russia who are lucky enough to get the early shots against nCoV say they feel more secure in ... |
 | Bac Ninh to spend $8 mln to give Covid-19 vaccine to 300,000 prioritized citizens Northern Bac Ninh Province would spend VND185 billion ($8 million) to inoculate around 300,000 prioritized residents against Covid-19 for free, the provincial People's Committee said ... |
Recommended
 Viet's Home
Viet's Home
Hanoi, South Africa Strengthens People-to-people Exchanges, Expands Multi-sector Cooperation
 Viet's Home
Viet's Home
Hue City to Raise Awareness on Mine Accident Prevention
 Focus
Focus
Vietnam Leaves Imprints on the World Peacekeeping Map
 Viet's Home
Viet's Home
“Global Vietnamese Singing 2025” - Connecting Hearts Longing for Homeland
 Viet's Home
Viet's Home
Vietnam’s People's Public Security Force Actively Contributes to UN Peacekeeping Operations
 Viet's Home
Viet's Home
HAUFO Enhances Competence of People-to-People Diplomacy Personnel
 Viet's Home
Viet's Home
Hands that Reserve Da Long Brocade Craft
 Viet's Home
Viet's Home