Covid-19 vaccines officially allowed to be imported into Vietnam
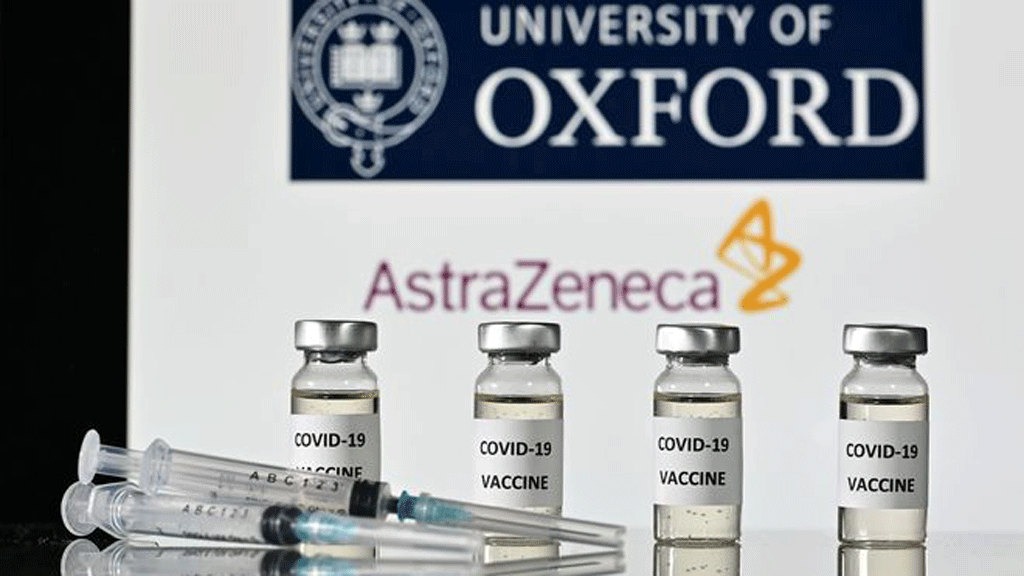 |
| Vietnam has 30 million doses of Covid-19 imported in this year's first 6 months. Photo: Thanhnien |
This morning, Minister of Health Nguyen Thanh Long signed a document approving the conditioning vaccine due to the urgent need to prevent and control the Covid-19 epidemic.
According to the Ministry of Health (MoH), the Covid-19 vaccine approved (AstraZeneca Covid-19 vaccine) is produced by AstraZeneca Company.
The vaccine is packed in boxes of 10 vials, each containing 8 doses, each dose of 0.5ml.
According to the MoH, in this first quarter, the first 50,000 doses of vaccine will arrive in Vietnam. There will be 30 million doses of vaccine to be provided to Vietnam in the first 6 months of this year, instead of until the year-end of original plan.
This is a large amount can be bought by Vietnam, after a time of negotiation while the demand for vaccines is very high in other countries. The Department of Science and Technology of the Ministry of Health will be the focal point in implementing clinical assessment (test injection on volunteers) to evaluate the safety and immunogenicity of Covid-19 vaccine AstraZeneca.
Imported vaccine batches made in Italy, UK and Germany will be imported and distributed in Vietnam by Vietnam Vaccine Joint Stock Company (VNVC).
The representative of the vaccine importer (Vietnam Vaccine Joint Stock Company - VNVC) said: “The Government and the Minister of Health have paid close attention for the Covid-19 vaccine to be imported into the country soon. The Evaluation and Licensing Council for vaccines in circulation has worked very urgently and strictly, proposing a license as soon as the vaccine is confirmed to meet standards. Along with that, the Drug Administration of Vietnam (Ministry of Health) and related agencies have joined in very urgently, with the effort for Vietnamese people to access vaccines as soon as possible ”.
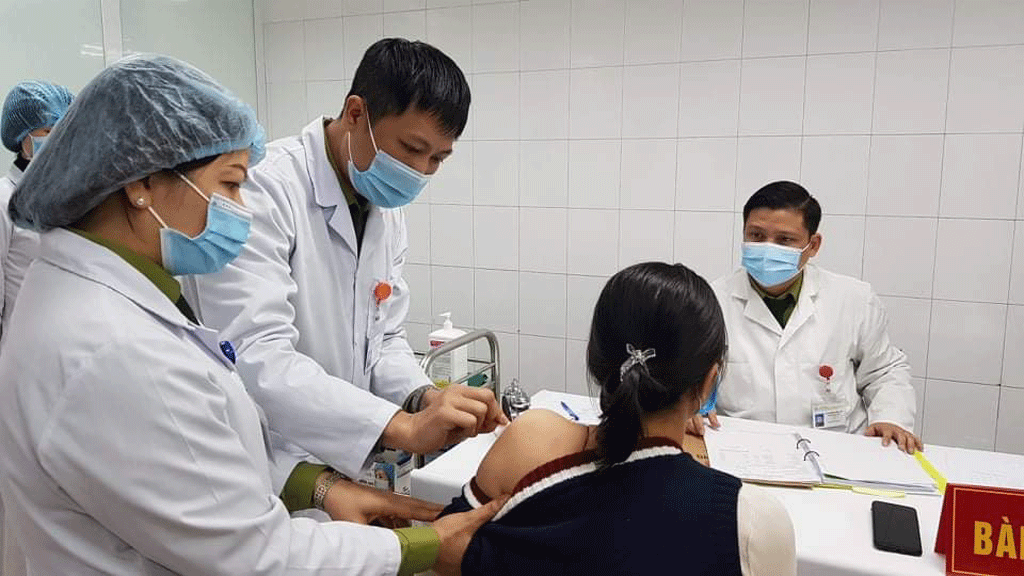 |
| A clinical trial will be implemented to assess the safety and immunogenicity of the imported Covid-19 vaccine. Photo: Thanhnien |
“During the negotiation phase, having vaccines bought was quite stressful due to great global demand. We would miss the first batch of vaccine if we were only a little bit late", said the representative of the importer. Up to now, VNVC has prepared an online software system for registration of Covid-19 vaccination upon arrival in Vietnam. Human resources for vaccination, cold storage system, and equipment for storing and distributing Covid-19 vaccines established, strictly followed the standards set by the Ministry of Health and recommended by the manufacturerto improve and maintain the stable quality of vaccines, in order to achieve maximum effectiveness for the vaccinated person.
Positive information for Vietnamese people
In the situation of Covid-19 epidemic outbreak in Vietnam recent days, there have been many cases confirmed positive to the Covid-19 in the community, especially in Hai Duong and Quang Ninh provinces, made people worried. Therefore, the information about Astra Zeneca's Covid-19 vaccine first batch imported into Vietnam is positive news for all Vietnamese people in preventing the Covid-19 pandemic.
 |
| Astra Zeneca's Covid-19 vaccine is injected into the body. Photo: AFP |
Many young people are also very interested in the information that Astra Zeneca Covid-19 vaccines will be imported into Vietnam in the near future. Quach Khanh My, a student at Can Tho University of Medicine and Pharmacy, shared: “It is hoped that Astra Zeneca Covid-19 vaccine batches will be imported into Vietnam in February on schedule to have vaccinations for people to avoid this extremely dangerous Covid-19 disease”.
Nguyen Thi Lan Anh, a student at the University of Technology of Ho Chi Minh City, said: “During this year, I always hope that the world will soon have a vaccine to prevent Covid-19 disease. Because I have never witnessed the harmful effects of such a terrible disease. Not only does it endanger human health, but the Covid-19 epidemic also brings with it other difficulties, especially economic difficulties and disruption to schooling. So, the news that Vietnam is about to have the first batch of Astra Zeneca's Covid-19 vaccine imported from the UK, really interested me. I just hope that when there is vaccine, we will effectively repel the Covid-19 epidemic and bring a peaceful life to the community".
Vaccination are given priority to fighters at the front line against the Covid-19 epidemic
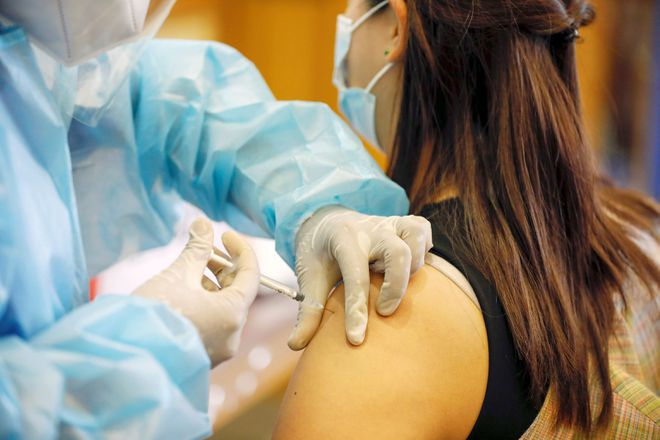 |
| Vaccination against epidemic Covid-19 REUTERS |
As a long-time employee in the medical industry, Dr. Tran Van Khanh, Director of the District 2 Hospital, Ho Chi Minh City, shared: “I am myself very happy when the Party, State and the Ministry of Health care much of Vietnamese people's health, especially the country's policy to support the entire cost of medical isolation and testing even though our country is still in economic difficulties. The purchase of the Covid-19 vaccines for the public is a very positive signal to prevent effectively the Covid-19 epidemic”.
As per Dr. Tran Van Khanh's suggestion, for the first Astra Zeneca Covid-19 vaccine batch imported in a limited quantity, the injection should be given to high-risk subjects including soldiers and medical staffs participating in the front line against the Covid-19 epidemic, people in epidemic provinces such as Quang Ninh, Hai Duong ...
 | Thousands of volunteers to be injected with Vietnam's first Covid-19 vaccine Leaders of the Ministry of Health said that 10,000 to 30,000 volunteers are expected to participate in the third phase of human trials of Nanocovax, ... |
 | All three Vietnamese volunteers injected with Covid-19 vaccines healthy After three days vaccinated with the Nano Covax vaccine, three Vietnamese volunteers are in stable health conditions with no unusual reactions. |
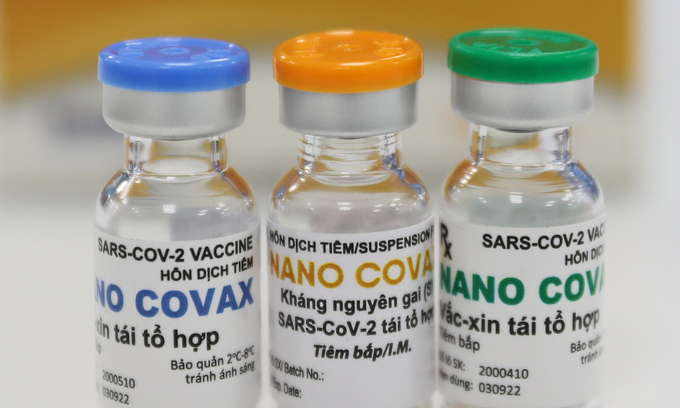 | Vietnam to conduct COVID-19 vaccine trial on willing volunteers Vietnam is currently recruiting willing volunteers for phase 1 COVID-19 vaccine clinical trial, with the injection is slated to be conducted starting December 17. |
Recommended
 National
National
Shangri-La Dialogue 22: Vietnam Highlights Some Issues of Ensuring Stability in a Competitive World
 National
National
Vietnam News Today (Jun. 3): PM Pham Minh Chinh to Attend UN Ocean Conference, Visit Estonia, Sweden
 National
National
Vietnam News Today (Jun. 2): Vietnamese Trade Mission Sounds Out Business Opportunities in United States
 National
National
Vietnam News Today (Jun. 1): Vietnamese, Japanese Firms Foster Partnership
 National
National
Vietnam News Today (May 31): Vietnam Strongly Supports Laos’s National Development
 National
National
Vietnam News Today (May 30): Vietnam, Venezuela Reinforce Ties Through People-to-people Diplomacy
 National
National
Vietnam News Today (May 29): Vietnam and Hungary to Expand Cooperation into New Areas
 National
National