Vietnam will take initiative in COVID-19 vaccine availability
 |
| Deputy Health Minister Tran Van Thuan speaking at Hanoi Medical University on March 15 (Photo: Thoi Dai) |
“We are having high hope on Covivac and Nonocovax, the two made-in-Vietnam vaccines currently under human trials. They are the fruits of Vietnamese researchers. Pre-clinical results in Vietnam, India, and the US show their efficacy on not only the common strain but also new variants in the UK and South Africa”, Tran Van Thuan, Deputy Minister of Health said March 15 morning at Hanoi Medical University. “If the two vaccines are approved, Vietnam could take the initiative in local COVID-19 vaccine. We might even be able to export the vaccines”.
Thuan further stressed that the ongoing clinical trials must be conducted in line with protocols, including close health monitoring post-injection. The entire implementation research and evaluation throughout phase 1 will be supervised by the Health Ministry. The Deputy Minister also tasked related units to follow three criteria: science – rigor – safety.
Vietnam on March 15 kicked off the first phase of human trials of the locally-produced Covivac vaccine at Hanoi Medical University, which involves 120 volunteers. The first 6 volunteers have been given either Covivac vaccine or placebo and were required to stay for further monitor at the medical facility within 24 hours.
The volunteers must first prove that they are physically eligible to get the vaccine, which involves survival indicators measurement, physical examination, screening, test sampling, pregnancy test (for females only). If all the ratios are satisfied, these people will wait for their turn in the waiting room.
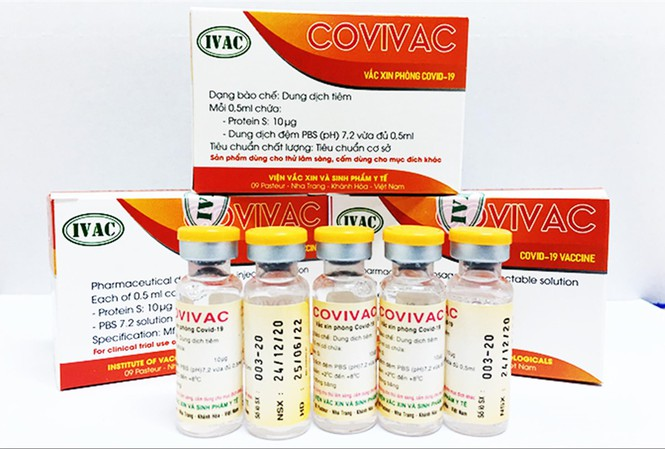 |
| Covivac is Vietnam's second homegrown COVID-19 vaccine to enter human trials (Photo: Tien Phong) |
Covivac is studied and developed by the Institute of Vaccines and Medical Biologicals (IVAC). It was proved effective on animals back in January. Clinical results in India, the US, and Vietnam showed the candidate was immunologically effective against the new coronavirus variants in the UK and South Africa.
The vaccine is expected to be launched later this year after passing through three phases of human trials. Moving forward, the IVAC plans to offer the Covivac vaccine at a price of VND60,000 (US$2.6) per dose.
Meanwhile, Nanogen's Nanocovax vaccine was Vietnam's first homegrown COVID-19 vaccine to enter human trials (starting December 2020). Nanocovax is priced at VND120,000 ($5.17) per dose.
Along with injections, Vietnam’s COVID-19 Nanocovax vaccine will also be developed in the form of eye-drop and nasal spray for special subjects.
The promising vaccine is currently under the second trial phase which involves 560 volunteers.
| Vietnam kicked off its vaccination campaign with AstraZeneca vaccine on March 8 in Hanoi, Hai Duong, Ho Chi Minh city. To date, 12 provinces and cities across the country have vaccinated its prioritized front-liners. Nearly 16,000 people have received the first dose.
Northern Hai Duong Province confirmed two Covid-19 cases from its ongoing outbreak Tuesday morning, raising its tally to 719 since late January. Both the new patients had close contact with previously confirmed cases. They have all been isolated as per the contact tracing protocol before the tests arrived as positive on Monday. Ever since community transmission returned to Vietnam on January 28, epicenter Hai Duong has recorded as many as 719 infections. From 6 am to 6 pm on March 15, Vietnam recorded three new cases of COVID-19, all Vietnamese citizens returning from abroad and being quarantined, according to the Ministry of Health. The remaining people are divided into 5 groups, with one expected to get placebo, the four others injected with different doses. The second dose is scheduled for the next 28 days. |
 | All COVID-19 vaccinated people in Vietnam are in stable health condition COVID-19 vaccinated people are all “in stable condition after one day of injection”, Director of the National Institute of Hygiene and Epidemiology Dang Duc Anh ... |
 | Vietnam continues vaccination campaign with AstraZeneca vaccine Given there is no blood clots in injected people after five days into the vaccination program, Vietnam continues to give prioritized groups the imported AstraZeneca ... |
 | In Photo: First medical workers in Da Nang vaccinated with Covid-19 Da Nang held the first injections of Covid-19 vaccines for medical staff on the morning of March 12. |
Recommended
 National
National
PM to Depart for Official Visit to Malaysia, Attendance at 46th ASEAN Summit
 National
National
Vietnam News Today (May 23): Vietnam–France Comprehensive Strategic Partnership Opens New Horizons for Cooperation
 National
National
Vietnam News Today (May 22): Stronger Vietnam-Israel Cooperation Expected in Science, Innovation and Labor
 National
National
Vietnam News Today (May 21): Vietnam Attends UN Commission on Crime Prevention and Criminal Justice's 34th Session
Popular article
 National
National
Deep Affection of International Friends
 National
National
Vietnam News Today (May 20): Hanoi Named Top Cultural, Artistic Destination in Asia
 National
National
Vietnam News Today (May 19): Norway Hails Vietnam’s Continued Emphasis on Upholding International Law
 National
National